
Total joint replacements (TJRs) were similarly distributed across all three treatment groups (6.7%–7.8%)’ (p, 1). The investigators reported that ‘rapidly progressive osteoarthritis (RPOA) was observed in 1.4% (4/283) and 2.8% (8/284) of patients in the tanezumab 2.5 mg and tanezumab 5 mg groups, respectively and none receiving placebo. The abstract emphasised more serious adverse events including rapidly progressive OA and joint replacement. The adverse effects of tanezumab were quantified in a variety of ways.
The large number of outcomes tested leads to inflated type I error rates and questions if results were chosen based on the significance.
Free clinical trials for osteoarthritis of the hip trial#
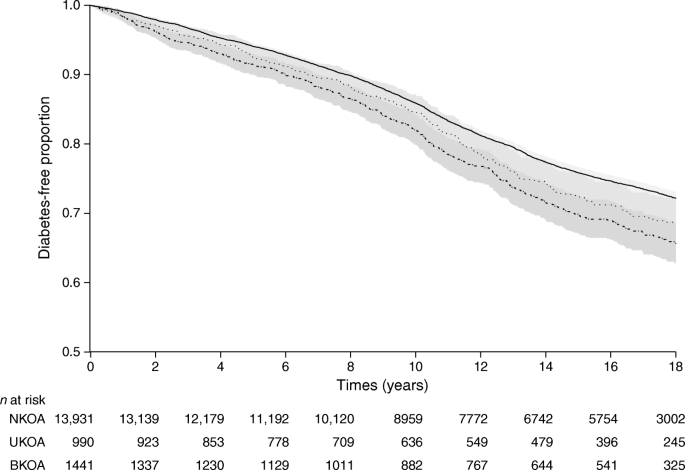
We found no discussion of the potential clinical impact of these findings in the paper.Įffects on six secondary outcomes were reported, but our concern with these estimates was that the trial registry listed 59 secondary outcomes with most of these including multiple measures compared over multiple time points. The findings indicate the expected difference is smaller than a patient shifting a response on a single item by one point. Given that the WOMAC Pain Scale ranges from 0 to 50 points and the WOMAC Physical Function Scale ranges from 0 to 170, changes of 0.71 points or less are, in our view, clinically irrelevant. For WOMAC Pain, mean differences were 0.61 or less compared with placebo, for WOMAC Physical Function, 0.71 points or less compared with placebo and for the global rating measure, 0.18 points or less. Mean differences among the three arms was less than one point for all primary outcomes. Treatment effects on primary outcomes were assessed by comparing changes from baseline to a 24-week follow-up.

The conclusion was ‘tanezumab 5 mg statistically significantly improved pain, physical function and PGA-OA, but tanezumab 2.5 mg only achieved two co-primary end points’ (p. For tanezumab 2.5 mg, there was a statistically significant improvement in WOMAC Pain and Physical Function, but not PGA-OA’ (p. The abstract reported the following results: ‘At week 24, there was a statistically significant improvement from baseline for tanezumab 5 mg compared with placebo for WOMAC Pain (least squares mean difference ±SE –0.62☐.18, p=0.0006), WOMAC Physical Function (–0.71☐.17, p<0.0001) and PGA-OA (–0.19☐.07, p=0.0051). The third primary outcome was the five-item Patient Global Assessment of OA with scores ranging from 1 to 5 with higher scores equating to worse self-reported symptoms and activity limitations. 6 Primary outcomes of the 2020 trial 4 were the WOMAC Pain Scale (numeric rating version with scores ranging from 0 to 50 with higher scores equating to more severe pain with activity) and the WOMAC Physical Function Scale (scores ranging from 0 to 170 with higher scores equating to more difficulty with activity). 4 This trial follows two similarly designed trials conducted on persons with symptomatic hip OA 5 or knee OA. 3 We believe the recently published trial by Berenbaum and colleagues 4 meets our secondary definition of publication spin and does not meet the recommendation endorsed by the ASA.īerenbaum and colleagues conducted a three-arm phase III randomised clinical trial of two doses of subcutaneously delivered tanezumab (2.5 vs 5 mg) as compared with placebo, applied to participants with symptomatic hip or knee osteoarthritis (OA).
2 A later editorial more explicitly states that conclusions not be based solely on statistical significance. The American Statistical Association (ASA) has commented on this issue, stating a statistically significant effect does not inform its size or importance. 1 In our view, a secondary but clinically important alternative type of publication spin is reliance on statistically significant findings without regard to potential clinical implications of the estimated effects. Publication spin, in the context of randomised clinical trials, is defined as ‘use of specific reporting strategies, from whatever motive, to highlight that the experimental treatment is beneficial, despite a statistically nonsignificant difference for the primary outcome, or to distract the reader from statistically nonsignificant results’ (p.

